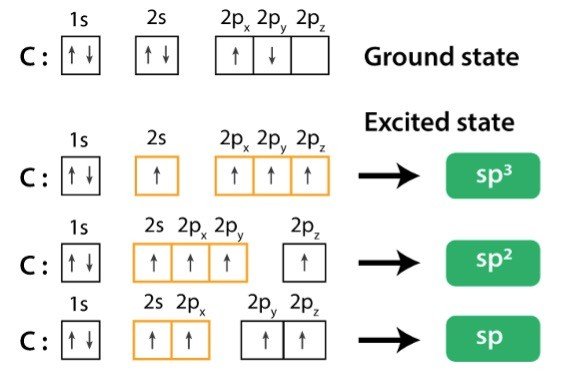
Therefore, in this case power of the hybridization state of N = 4-1 = 3 i.e. So, in NH 3 there is a total of four σ bonds around central atom N. three sigma (σ) bonds and one lone pair (LP) i.e. In NH 3: central atom N is surrounded by three N-H single bonds i.e. In addition to these each lone pair (LP) and Co-ordinate bond can be treated as one σ bond subsequently.Ī. Power on the Hybridization state of the central atom = (Total no of σ bonds around each central atom -1)Īll single (-) bonds are σ bond, in double bond (=) there is one σ and 1π, in triple bond (≡) there is one σ and 2π. S + p (1:1) - sp hybrid orbital s + p (1:2) - sp 2 hybrid orbital s + p (1:3) - sp 3 hybrid orbitalįormula used for the determination of sp, sp2 and sp3 hybridization state: We Know, hybridization is nothing but the mixing of orbital’s in different ratio to form some newly synthesized orbitals called hybrid orbitals. Prediction of sp, sp 2, sp 3 Hybridization state
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
